Parascript reports an outstanding year in sales growth with continued product innovation, new partnerships and high-profile client wins.


Parascript reports an outstanding year in sales growth with continued product innovation, new partnerships and high-profile client wins.

Parascript will sponsor, speak, and demonstrate its “Trend-Setting Product for 2015” at KMWorld Booth 301 in Washington, D.C.

WesTech shares its new AP automation solution at Parascript’s booth showcasing the Parascript and M-Files integration at M-Files 2015 Partner Conference

Document Imaging Report breaking news on Parascript focuses on how contest helps Parascript explore the classification market.

KMWorld names Parascript in its Trend-Setting Product for 2015 List after panelists reviewed over 200 vendors and 1,000 separate product offerings.

CU Times article by Greg Council addresses document management challenges, identifies core problems and potential solutions.

New version of EDAC’s ReadSCRIPT Advanced Handwriting Recognition Module powered by Parascript

Find out how Parascript transforms how businesses manage document-centered work through automation.

One qualifying business will receive Parascript’s document classification solution if selected in the Parascript Ultimate Document Challenge.

Document Imaging Report interviews Parascript about its new document classification software aimed at E-Governance.

CUToday published key tips to remaining successful in the mortgage business through contract automation powered by classification technology.
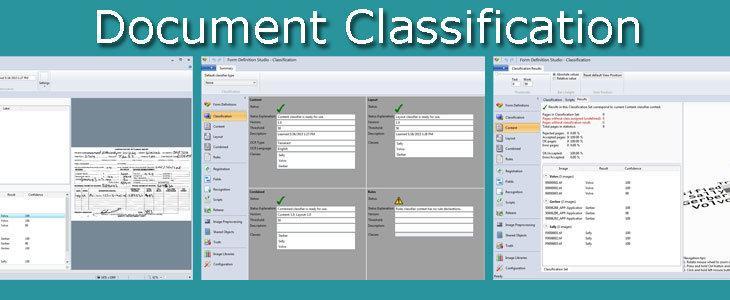
With Parascript Document Classification, business users directly leverage the efficiency and accuracy of advanced document auto-classification.